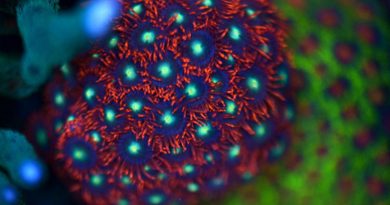
Dosing Trace Elements in Reef Tanks for more Colorful Acropora
Today we’re going to speak about trace elements and how to manage their levels in our tanks with DIY remedies. To begin, what is a trace element? It’s something that’s necessary for various biological processes, but only in trace levels. Trace levels are less than 1% by volume.
Concentrations of these many trace elements vary greatly in the ocean, depending on water depth and geographic location.
All we can do now is try to find a middle ground that works for our tanks. Some of these, such as iron and selenium, are hardly detectable in the natural ocean but are vital to the health of the organisms that dwell there. Dose what you haven’t tested. I can’t emphasize how crucial it is not to dose stuff that you aren’t paying attention to.
It’s especially crucial to have an accurate means to test for trace elements because some of them can be hazardous if there’s too much in the water.
Consider copper dosing: you’d never do that in a reef tank, yet our animals require trace amounts of it to thrive. Iodine is also vital, but at a larger dosage, it will disinfect your entire tank.
Let’s get started with iodine. Soft coral, shrimp, sponges, tunicates, gorgonians, and other organisms benefit from iodine. Remember that iodine is difficult to detect.
If you’re going to dose it, exercise caution and consider performing an ICP test. If you wish to produce your own dosage solution for it, potassium iodide is a good option. Another trace element is strontium, which is employed in our tanks similarly to calcium.
It’s found in the skeletons of growing things, and snails require it to grow their shells. Strontium chloride hexahydrate, for example, can be used to boost strontium levels in your tank. Bacteria and dinoflagellates that live inside the coral utilize molybdenum to flourish. Having molybdenum in our tank is also significant because these little organisms are crucial to the overall health of our fish and coral. You can raise the molybdenum levels in your tank water by using sodium molybdate.
Zinc is another trace element that is especially beneficial in biological processes. That are necessary for the overall health of the animals in our tanks. Zinc is very vital for the proper functioning of various enzymes in our animals. If you’re low on zinc, which isn’t a typical condition but can happen. Zinc chloride is a decent way to supplement it. Cells, particularly macroalgae cells, utilize manganese as they grow. In fact, macroalgae is capable of entirely removing free manganese from water.

Manganese sulfate can be used to provide manganese. There are other trace elements, such as selenium, gold, cobalt, and a whole host of others. That you don’t have to bother about dosing.
To obtain them in your tank water, all you have to do is change the water every now and then. Your salt combination has many of these fringe trace elements. If you dose those other things, the impurities in those compounds will add even more. Now consider the ingredients in your dosage chemical.
If you want to dose iodine, for example, potassium iodide is a useful source.
It does, however, contain both iodine and potassium. Potassium iodide has a molecular weight of around 166, and the formula is KI. Which means it has one potassium atom and one iodine atom. Because potassium has an atomic weight of 39 and iodine has an atomic weight of 126. Potassium iodide has far more iodine by weight than potassium. Because it contains potassium, you would not want to use it to elevate the potassium level in your tank. The iodine would kill everything.
Sulfates or chlorides of the metal that we want to increase are the most typical compounds we utilize for trace element dosage. It’s also not as harmful to add a lot of chloride, for example, to raise strontium using strontium chloride.
After all, the saltwater in our tank is saltwater because it contains a lot of sodium chloride. So, now that you’ve chosen to do it, where can you get these chemicals? Online, of course!
And one firm that I’ve seen mentioned in several forums is Alpha Chemicals. Although you can get these chemicals from anywhere: eBay, Amazon, etc. Be sure to buy something with a known purity, and obviously you want it to be as high as possible. The purity of trace elements isn’t that important because we’re only dosing a tiny amount of them in the first place.
So by the time you expand that out to your entire tank. There’ll only be a few micrograms of whatever impurity you have. So who cares if it’s.01 percent lead? Now, how do you figure out how much to dose. How do you figure out how much of these things to dose? It’s not difficult; all you have to do is do some basic math.
Then it’s only a matter of multiplying your own figures to get the equivalent concentration. Then following the Seachem instructions, for example. One part per million equals one microgram per milliliter, which is a handy conversion to remember.
And, because the metric system is incredible, one microgram per milliliter equals one milligram per liter. Remember, if you forget, simply go to Google and type in mg to ug to get the conversion.